AbstractPurposeThis study aimed to investigate the reliability of coagulation tests after storage of centrifuged samples in the primary collection tube with plasma remaining on top of the cells at room temperature and to compare results with current CLSI guidelines and previous studies.
MethodsSurplus blood samples from 20 patients were stored in the primary collection tube at room temperature, and were tested 2, 4, 6, 8, 12, 24, and 48 hr after the initial testing. The tests included aPTT/PT/fibrinogen/D-dimer, coagulation factors (F) VIII/IX/XI/XII/II/V/VII/X, von Willebrand factor (vWF) antigen and activity, antithrombin III (ATIII), fibrinogen degradation product (FDP), and dilute Russell’s viper venom time (dRVVT) screening and confirmation. A clinically significant difference was determined by a percentage change of > 10% according to the 99% confidence interval.
ResultsStorage of the sample in aPTT/PT/FVIII/FIX/FXI/FXII/FII/FV/FVII/FX/dRVVT screening and confirmation provided acceptable results up to 12/24/2/12/6/4/6/2/8/6/12, and 24 hr, respectively. For fibrinogen/D-dimers/vWF antigen and activity/ATIII/FDP, storage up to 48 hr also provided acceptable results. Compared to CLSI guidelines, the same acceptable storage periods for PT/FXII, shorter periods for FVIII/FV, and longer periods for the others tests were identified. Compared to previous studies that stored plasma after aliquotation, the acceptable storage periods were generally shorter for the coagulation factor assays.
ConclusionThe recommendations from the CLSI guidelines are too stringent in general but more strict storage limits should be applied to FVIII/FV. When additional coagulation factor assays are ordered and additional sample acquisition or preparation is not possible, more strict acceptable storage time criteria would be needed.
IntroductionStorage conditions, such as storage time and temperature, can affect the stability of coagulation factors and influence the results of coagulation assays [1]. Therefore, a clinical laboratory should determine the allowable time interval between collection of the specimen and testing of the sample stored at a certain temperature. The Clinical and Laboratory Standards Institute (CLSI) guidelines (H21-A5) recommend that whole blood samples or plasma samples stored at room temperature for routine hemostasis tests or determination of coagulation factors should be analyzed within 4 hr after sample collection, with the exception of prothrombin time testing with stability up to 24 hr [2].
However, there have been some studies suggesting samples stored for prolonged time periods are acceptable for reliable testing [3-6]. For example, there were no clinically relevant changes in prothrombin time (PT) test results with up to a 24–48 hr delay [4]. These data are practically important, because if a longer storage time were acceptable, resampling for additional coagulation testing and rejection of specimens due to prolonged delivery could be reduced. However, there are some differences between studies according to the storage conditions, testing method, and criteria for acceptability [5]. Most studies have dealt with unspun blood samples (focusing on time from sample collection to delivery to the laboratory) or separated plasma [4,5]. Therefore, these study data are not applicable to additional tests of samples with prolonged storage, considering most clinical samples are stored in the primary collection tube after initial testing. Additionally, there are few studies that include testing for fibrinogen degradation product (FDP) and coagulation inhibitors.
This study aimed to investigate the stability of coagulation tests after storage of centrifuged samples in the primary collection tube with plasma remaining on top of the cells at room temperature for different time periods and to evaluate whether a longer storage period is acceptable compared with current CLSI guidelines. In this study, along with routine coagulation tests such as activated partial thromboplastin time (aPTT), prothrombin time (PT), fibrinogen, D-dimer, coagulation factors VIII (FVIII)/IX (FIX)/XI (FXI)/XII (FXII)/II (FII)/V (FV)/VII (FVII)/X (FX), and von Willebrand factor antigen (vWF antigen) and activity (vWF activity), tests such as antithrombin III (ATIII), FDP, dilute Russell’s viper venom time screening (dRVVT screen) and confirmation (confirm) were conducted.
Methods1. Sample collection and processingThis study was approved by the Institutional Review Board of the authors’ institution (Ulsan University Hospital, approval number: UUH2019-12-014). Blood samples from 20 patients undergoing coagulation testing for preoperative evaluation were collected in 3.2% trisodium citrate tubes (BD Vacutainer, Ref. 363048; Becton Dickinson and Company, Franklin Lakes, NJ, USA). Centrifugation of the tubes took place within 30 minutes after blood collection. The samples were stored in the primary collection tube at room temperature (20–25℃) and were tested 2, 4, 6, 8, 12, 24, and 48 hr after the initial testing.
2. Test reagents and coagulation analysisTests were performed on the ACL TOP 500 (Instrumentation Laboratory Company, Bedford, MA, USA) instrument using HemosIL reagents (Instrumentation Laboratory Company) for aPTT (SynthASil), PT (RecombiPlasTin 2G), fibrinogen (Fib-C XL), D-dimers (D-Dimer HS), FVIII (Factor 8 deficient plasma), FIX (Factor 9 deficient plasma), FXI (Factor 11 deficient plasma), FXII (Factor 12 deficient plasma), FII (Factor 2 deficient plasma), FV (Factor 5 deficient plasma), FVII (Factor 7 deficient plasma), FX (Factor 10 deficient plasma), vWF Ag (vWF Antigen), vWF activity (vWF Activity), ATIII (Liquid AT), FDP (FDP), dRVVT screening (dRVVT Screen), and dRVVT confirmation (dRVVT Confirm).
3. Statistical analysisThe test results for each storage period were presented as the median with a corresponding interquartile range (IQR). Friedman repeated measure analysis of variance by ranks (Friedman test) was performed to check whether the storage period affected test results. Wilcoxon signed rank tests with Bonferroni adjustment, used to examine statistically significant differences compared to the initial result, were performed as a post hoc test, with the significance level set at 0.05.
The percentage change (%Changet=x) between the initial result (Resultt=0) and the results of subsequent storage periods (Resultt=x) was calculated using the following formula:
A percentage change value outside the interval [Q1-1.5× IQR,Q3 +1.5× IQR] was regarded as an outlier. The percentage change value excluding outliers was presented as the mean percentage change with a corresponding 99% confidence interval (99% CI) for each storage period using the t-distribution. A change compared with the initial result was considered acceptable when the 99% CI was within ±10%, and an initial measurement of > 10% according to the 99% CI was defined as a clinically relevant difference, which was identical to criteria applied to a previous study [4]. All statistical analyses were performed using R version 3.6.3. (R Foundation for Statistical Computing, Vienna, Austria).
ResultsThe coagulation assay test results from different storage periods are displayed in Table 1 with statistically or clinically significant differences indicated. The direction of change is schematically illustrated in Figure 1.
1. Identification of acceptable storage periods at room temperature for each testFor aPTT, the 99% CI of the percent change exceeded 10% after 24 hr storage (clinically significant), and an acceptable storage period at room temperature was determined as up to 12 hr. For PT, a clinically significant difference was seen after 48 hr storage and an acceptable storage period at room temperature was determined as up to 24 hr. Clinically significant differences in FVIII, FIX, FXI, FXII, FII, FV, FVII, and FX were seen after 4, 24, 8, 6, 8, 4, 12, and 8 hr storage, respectively, and acceptable storage periods at room temperature for these tests were determined as up to 2, 12, 6, 4, 6, 2, 8, and 6 hr, respectively. For these coagulation factors, the result values generally decreased along with prolonged storage, except for FXII with increased values after 24–48 hr storage. For fibrinogen, D-dimers, vWF antigen, vWF activity, ATIII, and FDP, no clinically significant differences were observed up to the 48 hr storage period, and an acceptable storage period at room temperature for these tests was determined as up to 48 hr. For dRVVT screening and confirmation, clinically significant differences were seen after 24 and 48 hr storage, respectively, and acceptable storage periods at room temperature for these tests were determined as up to 12 and 24 hr, respectively (Table 1).
2. Identification of storage periods with statistically significant differences for each test compared with initial test resultsThe Friedman test showed significant differences in all of the test items, including D-dimer (P= 0.040) vWF antigen (P= 0.006), and vWF activity (P= 0.036), which showed similar median values between different storage periods. For aPTT and PT, statistically significant differences by Wilcoxon signed rank test were seen after 8 and 24 hr storage, respectively. Statistically significant differences for FVIII, FIX, FXI, FXII, FV, FVII, and FX were seen after 2, 24, 12, 8, 12, 48, and 48 hr storage, respectively. Statistically significant differences in ATIII and FDP were seen after 8 and 24 hr storage. For dRVVT screening, a statistically significant difference was seen after 24 hr. For D-dimer, FII, fibrinogen, vWF antigen, vWF activity, and dRVVT confirmation, there was no significant difference between initial test results and those from different storage periods (Table 1).
3. Comparison of acceptable storage periods at room temperature in our study with those presented in CLSI guidelines and a previous studyCompared with CLSI guidelines, our study presented the same or longer acceptable storage periods in all test items, with the exception of both FVIII and FV [2 hr (our study) vs. 4 hr (CLSI guideline)], which are labile coagulation factors. Compared with a previous study [4] that applied the same criteria for acceptability (> 10% according to the 99% CI) but stored plasma after aliquotation, our study, which stored samples in the primary collection tube after centrifugation without aliquotation, revealed partially different results. Acceptable storage times from our study were similar for aPTT, PT, fibrinogen, D-dimers, FVIII, FV, vWF Ag, and vWF activity, and were shorter for FIX, FXI, FXII, FII, FVII, and FX compared to the previous study [4] (Table 2).
DiscussionOur study investigated the effects of centrifuged primary collection tube storage for 2, 4, 6, 8, 12, 24, 48 hr at room temperature on coagulation assays. Although many studies have evaluated the stability of samples for coagulation tests, they evaluated the effect of delayed storage with unspun blood samples, focusing on time from sample collection to its delivery to the laboratory [3,5,7,8]. Some studies have also evaluated the effect of delayed storage as separated plasma in a secondary tube [4-9]. Our study evaluated acceptable storage periods for various coagulation tests using the primary collection tube stored after initial centrifugation without separation or aliquotation of the primary sample. Given that additional coagulation tests are frequently ordered with the initially obtained, primary sample without aliquotation or separation of the primary tube in the clinical setting especially for outpatients, it can be expected that our study would give useful data about acceptable time limits for delayed or additional testing using the sample after an initial test stored in the primary collection tube after centrifugation, which could reflect clinical situations better than previous studies [4-9].
There are no unified guidelines for clinically acceptable bias criteria. Some studies have suggested a limit of mean percentage change greater than 10% to be a clinically significant difference [7,8]. Other studies have suggested a classification considering the distribution of changes: >10% in <25% of samples as a moderate effect and changes >10% in >25% of samples as a large effect [10]. Zürcher suggested the method considering the impact of test imprecision on the results, and we applied this method to our study [3]. This percent change of > 10% according to the 99% CI is also an arbitrarily chosen criteria, but it seems to be accepted by others and is useful as it provides data based on objective probability [4].
Compared with CLSI guidelines, our findings suggest the same acceptable time limits for PT and FXII, shorter limits for FVIII and FV, and longer limits for other tests. These results are similar to a previous study [4], and therefore, except for both FVIII and FV, which are well-known labile coagulation factors, it can be suggested that the CLSI guideline recommendations are too stringent, but more stringent storage limits should be applied to both FVIII and FV. In addition, when compared to a previous study [4] we also found that the acceptable storage times from our study were generally shorter for the coagulation factor assays and were similar for the other assays. These results suggest that when additional coagulation factor assays are ordered and additional sample acquisition or preparation is not possible, more strict acceptable storage time criteria would be needed compared to those reported in the previous study [4]. An increase in FXII activity was observed in our study after a storage time of 24 hr and the previous study [4] reported similar results. This phenomenon might be due to contact activation, and in our study, the change was more dramatic with a marked decrease up to 12 hr and a sharp increase after 24 hr. The prolonged contact with cells might affect the results, but we do not have a plausible explanation for this finding.
In our study, although statistically significant differences were observed, some biases were still within an acceptable limit. For example, the result of aPTT at 8 hr was statistically significantly different from the result at 0 hr, while it was within the acceptable storage period. On the other hand, the storage periods of 8 hr for FXI, 6 hr for FXII, 8–48 hr for FII, 6–8 hr for FV, 12–24 hr for FVII, and 8–24 hr for FX showed results beyond the acceptable storage period but did not show statistically significant differences compared to results at 0 hr. This finding could be affected by result dispersion, which might reflect intra-laboratory imprecision and sample variation, but this explanation should be confirmed in a more comprehensive future study.
In conclusion, our study findings suggest the same acceptable storage periods at room temperature for PT and FXII, shorter storage times for FVIII and FV, and longer storage times for the other tests compared with CLSI guidelines (H21-A5 2008). Compared to a previous study that stored plasma after aliquotation, our study found that the acceptable storage periods were generally shorter for the coagulation factor assays and were similar for the other assays. Therefore, it can be suggested that the recommendations from the CLSI guidelines are too stringent, with the exception of both FVIII and FV for which more strict storage limits should be applied. When additional coagulation factor assays are ordered and additional sample acquisition or preparation is not possible, more strict acceptable storage time criteria would be needed.
NotesAuthors’ contributions Hyun-Ki Kim analyzed the data, and drafted the manuscript. Ji-Hun Lim and Joseph Jeong analyzed the data and interpreted the results. Seon-Ho Lee contributed to the conception of the project. Sang Hyuk Park designed and supervised the study. All authors reviewed the manuscript and provided critical feedback. Fig. 1.The test results of coagulation assays according to the prolonged storage (2–48 hours) periods. Outliers excluded for confidence interval calculations are marked as “X”. The results from the same sample are connected with a line.
aPTT, activated partial thromboplastin time; PT, prothrombin time; vWF Ag, von Willebrand factor antigen; vWF activity, von Willebrand factor activity; FDP, fibrinogen degradation product; dRVVT screen, dilute Russell’s viper venom time screening; dRVVT confirm, dilute Russell’s viper venom time confirm.
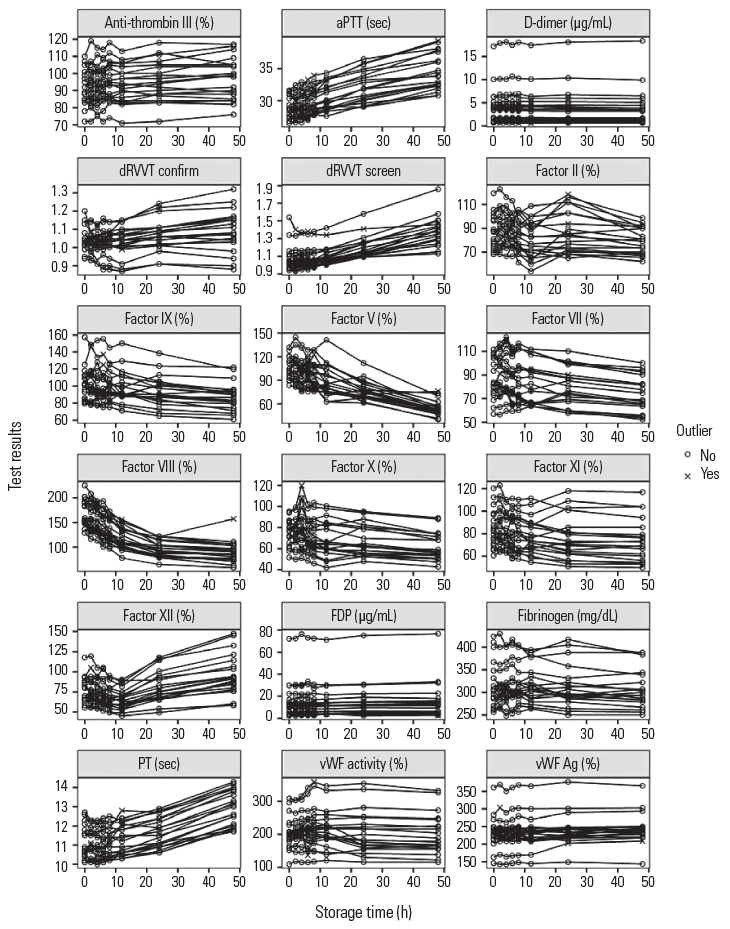
Table 1.The coagulation test results from different storage periods
* denotes a statistically significant difference (P < 0.05) compared with the initial results (time 0 hr) using Wilcoxon signed rank tests with Bonferroni adjustments; † denotes a clinically significant difference defined by 99% CI for the percent change exceeding 10%. n, number of samples; hr, hours; aPTT, activated partial thromboplastin time; PT, prothrombin time; F, factor; vWF Ag, von Willebrand factor antigen; vWF activity, von Willebrand factor activity; ATIII, antithrombin III; FDP, fibrinogen degradation product; dRVVT screen, dilute Russell’s viper venom time screening; dRVVT confirm, dilute Russell’s viper venom time confirm; IQR, interquartile range; CI, confidence interval. Table 2.Comparison of the acceptable storage periods at room temperature according to the CLSI guidelines, previous study, and our study findings
* For aPTT and PT tests using uncentrifuged or centrifuged sample with plasma remaining on top of the cells, and for other tests using centrifuged sample. For aPTT using plasma containing unfractionated heparin, plasma should be removed within one hour of collection. h, hours; aPTT, activated partial thromboplastin time; PT, prothrombin time; F, factor; vWF Ag, von Willebrand factor antigen; vWF activity, von Willebrand factor activity; ATIII, antithrombin III; FDP, fibrinogen degradation product; dRVVT screen, dilute Russell’s viper venom time screening; dRVVT confirm, dilute Russell’s viper venom time confirm. References1. Favaloro EJ, Lippi G, Adcock DM. Preanalytical and postanalytical variables: the leading causes of diagnostic error in hemostasis?. Semin Thromb Hemost 2008;34:612-34.
2. Adcock DM, Hoefner DM, Kottke-Marchant K, Marlar RA, Szamosi DI, Warunek DJ. CLSI Document H21-A5. Collection, transport, and processing of blood specimes for testing plasma-based coagulation assays and molecular hemostasis assays: approved guideline. 5th edition: The Clinical and Laboratory Standards Institute (CLSI) 2008.
3. Zürcher M, Sulzer I, Barizzi G, Lämmle B, Alberio L. Stability of coagulation assays performed in plasma from citrated whole blood transported at ambient temperature. Thromb Haemost 2008;99:416-26.
4. Linskens EA, Devreese KMJ. Pre-analytical stability of coagulation parameters in plasma stored at room temperature. Int J Lab Hematol 2018;40:292-303.
5. Toulon P, Metge S, Hangard M, Zwahlen S, Piaulenne S, Besson V. Impact of different storage times at room temperature of unspun citrated blood samples on routine coagulation tests results. Results of a bicenter study and review of the literature. Int J Lab Hematol 2017;39:458-68.
6. Rimac V, Coen HD. Is it acceptable to use coagulation plasma samples stored at room temperature and 4°C for 24 hours for additional prothrombin time, activated partial thromboplastin time, fibrinogen, antithrombin, and D-dimer testing?. Int J Lab Hematol 2017;39:475-81.
7. Salvagno GL, Lippi G, Montagnana M, Franchini M, Poli G, Guidi GC. Influence of temperature and time before centrifugation of specimens for routine coagulation testing. Int J Lab Hematol 2009;31:462-7.
8. Kemkes-Matthes B, Fischer R, Peetz D. Influence of 8 and 24-h storage of whole blood at ambient temperature on prothrombin time, activated partial thromboplastin time, fibrinogen, thrombin time, antithrombin and D-dimer. Blood Coagul Fibrinolysis 2011;22:215-20.
9. Feng L, Zhao Y, Zhao H, Shao Z. Effects of storage time and temperature on coagulation tests and factors in fresh plasma. Scientific Reports 2014;4:3868.
10. van Geest-Daalderop JH, Mulder AB, Boonman-de Winter LJ, Hoekstra MM, van den Besselaar AM. Preanalytical variables and off-site blood collection: influences on the results of the prothrombin time/international normalized ratio test and implications for monitoring of oral anticoagulant therapy. Clin Chem 2005;51:561-8.
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||