Remnant Lipoproteins: A Subfraction of Plasma Triglyceride-Rich Lipoproteins Associated with Postprandial Hyperlipidemia
Article information
Abstract
Plasma remnant lipoproteins, a subfraction of triglyceride (TG)-rich lipoproteins (TRLs), have been shown to be a causative factor for atherosclerosis and thrombosis. TRLs are rich in chylomicrons (CM), very low-density lipoproteins (VLDL) and their remnants (partially metabolized CM and VLDL). Remnant lipoproteins are significantly increased in the postprandial plasma and are predictive of the risk for coronary heart disease (CHD), independent of the total cholesterol, TG, LDL-C or HDL-C. The biochemical and physiological characteristics of remnant lipoproteins are very similar to that of oxidized LDL (ox-LDL), but the plasma concentrations of ox-LDL are extremely low compared with concentrations of remnant lipoproteins. The isolation of remnant lipoproteins from plasma has been difficult for a long time because ultracentrifugation and electrophoresis, the most common methods for the isolation of lipoprotein fractions, can’t specifically isolate remnant lipoproteins. The immuno-separation method enables specific isolation of remnant lipoproteins from plasma as an unbound fraction to the anti-apoB100 and apoA-1 immuno-affinity gel. Using this technique, an easy remnant lipoprotein-cholesterol assay (RLP-C) can be performed to investigate the clinical significance of remnant lipoproteins in the plasma. Over the last two decades, this immune-separation technique has provided new insights into remnant lipoproteins.
Introduction
Remnant lipoproteins (a subfraction of triglyceride [TG]-rich lipoproteins) have received a lot of attention recently as a risk factor for cardiovascular disease. Dr. Borge Nordestgaard of the University of Copenhagen, Denmark, refers to remnant lipoprotein-cholesterol as “ugly cholesterol”, similar to the way low-density lipoproteins (LDL)-C is considered “bad cholesterol”. It has been difficult to isolate remnant lipoproteins, specifically from TG-rich lipoproteins, because chylomicron (CM) and very low-density lipoproteins (VLDL) remnants are found at a similar density, charge and particle size with nascent CM and VLDL, as shown in Fig. 1. Also, the most important clinical feature of remnant lipoproteins is the association with postprandial metabolism of TG-rich lipoproteins. We have clarified that approximately 80% of the postprandial increase of TG is composed of TG in remnant lipoproteins. Therefore, it is most important to pay attention to postprandial hyperlipidemia when discussing the clinical significance of plasma remnant lipoproteins.

Major classes and subclasses of plasma lipoproteins. There are many subclasses of lipoproteins in CM, VLDL, LDL, and HDL, which have different biological and chemical characteristics. Remnant lipoproteins are found in the CM and VLDL fraction with similar particle sizes and charges to nascent ones. Ox-LDL and small dense LDL are found in the LDL fraction.
Plasma triglycerides are surrogates for TG-rich lipoproteins (TRL) and are rich in chylomicrons (CM: apoB48 carrying particles), very low-density lipoproteins (VLDL: apoB100 carrying particles) and their remnants (partially metabolized CM and VLDL). TRLs and their remnants are significantly increased in the postprandial plasma and are predictors of the risk of coronary heart disease (CHD)[1,2], independent of the total cholesterol, TG, LDL or HDL cholesterol level. Recently, non-fasting TG levels were shown to be significant risk indicators for CHD events[3-5]. Zilversmit[6] first proposed in 1970 that postprandial hyperlipidemia reflects the increased CM remnants as the most common risk factor for atherogenesis in individuals without familial hyperlipoproteinemia. The concept that postprandial CM and CM remnants are a major cause of common atherogenesis is supported by evidence that CM is significantly increased in the intestine after food intake, and a large amount of CM flows into the blood stream through the thoracic duct. Therefore, CM and CM remnants appear to be major lipoproteins in the postprandial hyperlipidemia. In addition, approximately 80% of the postprandial increase in triglycerides are accounted for by the apoB-48-containing lipoproteins (CM or CM remnants) until recently[7]. Therefore, large quantities of triglycerides are believed to be transported by a very small number of apoB-48-carrying CM particles. This has been a controversy for a long time because of the lack of methods for isolation and detection of remnant lipoproteins.
To investigate the characteristics of remnant lipoproteins, we developed an immuno-separation method that enables the isolation of remnant lipoproteins from plasma as remnant-like lipoprotein particles (RLP)[8,9], and we have used this method to investigate the composition and particle size of CM remnants and VLDL remnants in postprandial plasma. This method provides a tool to isolate both CM and VLDL remnants from plasma simultaneously as RLPs, which reflect the typical biochemical characteristics of CM and VLDL remnant lipoproteins previously reported[1,2]. Also, the further separation and detection of lipoprotein subclasses by a gel permeation highperformance liquid chromatographic (HPLC) system[10] and the detection method of apoB-48 in fractionated lipoproteins by a highly specific ELISA[11] have enabled the investigation of remnant lipoproteins.
In the last part of this review article, we describe the therapeutic effectiveness of the different kinds of drugs that reduce the plasma level of remnant lipoproteins.
Metabolism of CM, VLDL, and their remnants in plasma
Fig. 2 shows the metabolic pathway of CM and VLDL. CM is secreted by the intestine after fat consumption. CM particles contain apoB-48 as a structural protein, which in humans is formed exclusively in the intestine after tissue-specific editing of the apoB-100 mRNA[12,13]. It appears that apoB-48-containing particles are continuously secreted from enterocytes, and at times of excessive triglyceride availability, lipid droplets fuse with nascent lipoprotein particles, resulting in the secretion of enormous chylomicrons[14,15]. Once the CM particle reaches the plasma compartment, apo A-I dissociates very rapidly[16] and acquires apoCs, in particular apoC-II, to enable efficient unloading of its massive triglyceride content after binding to the lipoprotein lipase (LPL), which is bound to the endothelium[17]. High-density lipoproteins (HDL) are a major reservoir for apoCs and apoE, but only in conditions with low HDL concentrations (found most often in hypertriglyceridemic subjects). The halflife of CM triglycerides in healthy subjects is very short, approximately 5 min[18]. The half-life of CM particles has been very difficult to estimate due to the difficulty in achieving adequate labeling of CM. The CM particle half-life is certainly longer than that for CM triglycerides and seems to be quite heterogeneous. Certain pools of CM remnants have a very long residence time, at least as long as similar-sized VLDL particles[19,20]. Furthermore, a major proportion of the CM remnants leave the plasma compartment quite rapidly, but are still quite large, i.e., 75 nm in diameter[19].

Metabolic pathway of CM, VLDL and remnant lipoproteins. After fat intake, the intestine secretes chylomicrons (CM), the triglycerides that are lipolyzed by lipoprotein lipase (LPL). The LPL reaction constitutes the initial process in the formation of TRL remnants. The VLDL secretion process is partly regulated by the rate of FFA influx to the liver. VLDL triglycerides are lipolyzed by endothelial-bound lipoprotein lipase and VLDL remnant particles are formed. The final TRL remnant composition is modulated by the cholesteryl ester transfer protein (CETP) reaction with HDL, hepatic lipase (HL), and the exchange of soluble apolipoproteins such as C-I, C-II, C-III and E. The great majority of the remnants are removed from plasma by receptor-mediated processes and the principal receptors are the LDL receptor and the LDL-receptor-related protein (LRP).
There is competition for lipolysis; CM and VLDL mix in the blood and the two TRL species compete for the same lipolytic pathway[20,21]. It has been shown that endogenous TRLs accumulate in human plasma after fat intake and the mechanism behind this phenomenon is explained by the delayed lipolysis of the apoB-100 TRL particles due to competition with CM for the sites of LPL action[21]. Similarly, endogenous TRLs accumulate in rat plasma due to competition with a CM-like triglyceride emulsion for the common lipolytic pathway[22]. The increase in the number of TRL apoB-100 particles is actually far greater than that of the apoB-48-containing lipoproteins in the postprandial state[23]. Of note, the accumulation of large TRL apoB-100 particles seems to be a sign in hypertriglyceridemic patients with coronary artery disease (CAD) compared with healthy hypertriglyceridemic subjects, suggesting a link between the accumulation of large VLDL and the development of atherosclerosis[24].
VLDL particles are secreted continuously from the liver (Fig. 2). In contrast to CM and its remnants, they are characterized by their apoB-100 content. The secretion of VLDL is under complex regulation, as the larger and more triglyceride-rich VLDL species are under strict insulin control in a dual sense. First, a number of more or less insulin-sensitive mechanisms regulate the availability of triglycerides for VLDL production. The free fatty acids (FFA), which are generated by lipolysis in adipose tissue through the action of hormone-sensitive lipase, provide a major source for hepatic VLDL secretion. Insulin stimulates the endothelial expression of LPL, the key enzyme in TRL metabolism, in a post-transcriptional manner[25,26]. Hepatic uptake of poorly lipolyzed VLDL or CM remnant particles may also contribute to the hepatocellular triglyceride availability. Similarly, reduced uptake of FFA in adipose and muscle tissues after LPL-mediated lipolysis of CM and VLDL shunts FFA to the liver[27]. Finally, the liver has the capacity for de novo synthesis of triglycerides and VLDL.
In contrast, the metabolic pathway of VLDL by hepatic triglyceride lipase (HTGL) seems to still be controversial because of the difficulties with measurement. HTGL has been reported to metabolize comparatively small remnant lipoproteins, although to a lesser extent than LPL. However, our recent studies have shown no correlation between HTGL activity and plasma TG, RLP-C, RLP-TG or even small dense LDL-C concentration in humans[28], although we did find a strong inverse correlation between LPL activity and these three parameters in both the fasting and postprandial state. Previous studies proposing a role of HTGL in remnant metabolism have been mainly based on animal studies using anti-HTGL antibodies in monkeys and rats, and have demonstrated an accumulation of remnant lipoproteins in plasma after HTGL-specific antibody treatment[29,30]. The demonstration that small dense LDL (sd LDL) is positively correlated with TG and remnant lipoproteins in plasma supports the concept that remnant lipoproteins are the precursor of sd LDL and are metabolized in the same pathway by LPL[28]. Based on these data, HTGL does not seem to play a significant role in the metabolic pathway of remnant lipoproteins, in contrast to findings from previous reports[29-33], but rather, plays a definitive role in HDL metabolism in humans.
Biochemical characteristics of CM and VLDL remnants
TRL remnants are formed in the circulation when apoB-48-containing CM of intestinal origin or apoB-100-containing VLDL of hepatic origin are converted by lipoprotein lipase into smaller and denser particles[31-34], as shown in Fig. 2. Compared with their nascent precursors, TRL remnants are depleted of triglycerides, phospholipids, and apoCs (and apoA-I and apoA-IV in the case of CM), and are enriched in cholesterol esters and apoE[35,36]. They can thus be identified, separated, or quantified in plasma on the basis of their density, charge, size, specific lipid components, apolipoprotein composition and apolipoprotein immune-specificity[37]. Each of these approaches has provided useful information about the structure and function of remnant lipoproteins, and has helped to establish the role of TRL remnants in the pathogenesis of atherosclerosis. Accurate measurement and characterization of plasma remnant lipoproteins, however, has proven to be difficult for the following reasons: despite their reduced size and triglyceride content, they are difficult to differentiate from their triglyceride-rich precursors; due to their rapid plasma catabolism, they exist in plasma at relatively low concentrations; and since remnants are at different stages of catabolism, they are markedly heterogeneous in size and composition. TRLs are known to become progressively smaller, denser and less negatively charged as they are converted into TRL remnants. They gradually lose triglycerides and, in relative terms, become enriched with cholesteryl esters.
Remnant lipoproteins are thus structurally and compositionally diverse, which has made it necessary to develop a variety of specific biochemical techniques for the detection, quantification and characterization of these lipoproteins. In light of such difficulties, we developed a new immuno-separation approach to separate a variety of remnant lipoproteins from the normal apoB100 (nascent VLDL and LDL)-carrying lipoproteins. A specific anti-apoB-100 antibody that does not recognize the α-helix structure of the apoB-51 region was developed and used for the isolation of the apoE-rich VLDL remnants[9].
Comparison between remnant lipoproteins and oxidized LDL (Ox-LDL) on atherogenesis
Table 1 shows the very high similarity in biochemical characteristics between remnant lipoproteins and Ox-LDL. Remnant lipoprotein can be identified as a kind of oxidized lipoprotein and its atherogenicity may be based on the similar biochemical properties of Ox-LDL. Most of the physiological characteristics of RLP can be explained by the same experimental methods with Ox-LDL.

The similarity of pro-atherogenic and pro-inflammatory properties of Ox-LDL and RLP in in vitro studies
As for the oxidative modifications of lipoproteins other than LDL, the susceptibility to oxidation of remnant lipoproteins has been elucidated by many researchers using the RLP isolated by the immnoaffinity gel method[8,9]. Unlike Ox-LDL, RLPs do not require further modification in vitro to be atherogenic (RLPs are atherogenic per se). Although there has not been a specific report describing the characteristic of beta-VLDL (typical remnant lipoproteins) as highly oxidized lipoproteins, in vitro studies of beta-VLDL demonstrated active formation of foam cells[38] and cell cytotoxicity[39], revealing high similarity with Ox-LDL. Beta-VLDL or RLP may carry more apoE particles (positively-charged apolipoproteins) than non-RLP in VLDL[40-42] due to the negative charge on the particles following oxidative modification generated in the circulation. Both LDL or Ox-LDL and RLP are categorized as atherogenic lipoproteins in plasma based on many clinical studies. The Ox-LDL can be prepared in vitro by adding CuSO4 to LDL for heavily oxidized LDL[43] or FeSO4 to LDL for minimally modified LDL[44]. In contrast, it has been difficult to isolate remnant lipoproteins from TRL without modification. Using the isolation method of RLP, Doi et al.[45] reported the susceptibility of RLP to oxidation. The incubation of isolated human red blood cells (RBCs) with RLP and its extracted lipids converted the shape of RBCs to that of echinocytes, but co-incubation with antioxidants suppressed the RLP-induced RBC transformation, suggesting that they exerted oxidative damage on the RBC surface membrane. Studies with HPLC and the postcolumn chemiluminescence method showed that RLPs contain a substantial amount of phosphatidylcholine hydroperoxide, one of the typical oxidized phospholipid components in Ox-LDL[46]. Peroxidized phosphotidylcholine also impaired EDR and had echinocytogenic action, both of which were suppressed by N-acetylcysteine, an antioxidant. RLP isolated from the patients being treated with alpha-tocopherol, an antioxidant, had a lower level of phosphatidylcholine hydroperoxide (15% of the amount in non-treated patients), which was associated with a lack of inhibitory action on EDR and with lesser effect on RBC transformation. Oxidized damage caused by extracted lipid components in RLP, especially peroxidized phospholipids, causes the cell surface membrane to deteriorate and may be responsible for RLP-induced impairment of EDR. The biologically-active properties such as cell death did not change significantly when RLP was further oxidized to Ox-RLP by CuSO4 or modified to carbamylated and methylated RLP[47]. Further, Tamura et al.[48] demonstrated that isolated RLPs were extensively oxidized compared with total VLDL by thiobarbituric acid reactive substance (TBARS) detection. The oxidation levels of RLP isolated from 10 patients with diabetes mellitus, 10 patients with ischemic heart disease and 10 normal subjects were compared with that of total VLDL by measuring the levels of TBARS. The results indicated that RLPs were oxidized in vivo to a greater extent than total VLDL in all three groups. It was unlikely that intra-experimental artifacts including auto-oxidation of RLP occurred during the separation procedure of RLP from VLDL. However, these results also raise the possibility that a greater amount of TBARS also means higher content of unsaturated fatty acids such as arachidonic acids and DHA, which are also very susceptible to oxidation. The large amount of lysophosphatdylcholine in CM remnants, a constituent of RLP, was first reported by Scow and Egelrud in 1976[49]. CM remnants were prepared in vivo by injecting rat CM intravenously into functionally hepatectomized rats, and 3 hours later, blood was obtained from the rats and CM remnants were isolated by ultracentrifugation[50]. Lipoprotein lipase readily hydrolyzed phosphatidylecholine of chylomicrons to lysophosphatidylcholine both in vitro and in vivo. A significant increase in lysophosphatidylcholine in CM remnants was always observed with the increase in apoE. Therefore, the presence of lysophosphatidylcholine in CM remnants reflects the formation of remnant lipoproteins from CM. One of the major characteristics of Ox-LDL is the presence of lysophosphatidylcholine, which has been recognized to be an active component of the various proatherogenic and proinflammatory properties of Ox-LDL. Kawasaki et al.[51] reported that CM remnants prepared as above induced apoptosis in vascular endothelial cells, induced monocyte chemo-attractant protein-1 expression in vascular smooth muscle cells[52], and regulated early response factor-1 (Egr-1) in vascular smooth muscle cells[53]. These results reported for CM remnants were observed with similar concentrations of Ox-LDL and RLP in in vitro studies.
These results indicate that there is a high degree of similarity in oxidative modification between Ox-LDL and RLP. Table 1 summarizes the striking similarity for atherogenic effects of Ox-LDL[54-60] and RLP in in vitro studies.
Platelet aggregation by remnant lipoproteins
The additive effects of remnant lipoproteins on platelet aggregation were observed by Takada and his colleagues. Knöfler et al.[61,62] found a strong potentiation by RLP of ADP-induced platelet aggregation, although they were not able to detect any direct platelet stimulation by RLP alone.
Saniabadi et al.[63] reported on the platelet-aggregating effects of RLP. They found that the addition of RLP to whole-blood samples containing minimum lipoprotein remnants induced significant platelet aggregation; and these effects were observed around and well below the physiologic concentrations, namely, concentrations of 0.08-3.2 mg/dL of RLP-C with the peak aggregation at 0.32 mg/dL. Interestingly, platelet aggregation with RLP was observed only if there were RBCs, and aggregates were found on the surface of RBC. In contrast, other lipoproteins did not markedly affect platelets.
Yamazaki et al.[64] also reported that shear-induced platelet aggregation (SIPA) was significantly enhanced by RLP in whole blood, but not in platelet-rich plasma (PRP). This enhancing effect was not dose-dependent and was greatest at 10 μg TG/mL. P-selectin expression induced by shear stress was only enhanced by RLP at a concentration of 100 μg TG/mL in both WB and PRP, while generation of platelet-derived microparticle (PMP) induced by shear stress was only enhanced by RLP at a concentration of 100 μg TG/mL in WB. Aspirin inhibited only the enhancement of SIPA by RLPs, while cilostazol inhibited the enhancement of not only SIPA, but also p-selectin expression and PMP generation by RLPs.
Daily pattern of plasma TG and remnant lipoprotein concentrations and changes in plasma levels after a fat load
The plasma TG concentration fluctuates throughout the day in response to meals. Even if measured after a 10- to 12-hr overnight fast (as is normal clinical practice), TG levels vary considerably more than LDL and HDL cholesterol levels.
As non-fasting TG levels are now known to be a significant risk for CHD events[3-5], the analysis of postprandial lipoproteins, rather than the fasting state, has come to be recognized as more important. We reported that non-fasting TG correlated more strongly with remnant lipoproteins than fasting TG[65]. The correlations between postprandial TG and remnant lipoprotein concentrations were significantly more robust when compared with fasting TG vs. remnant lipoprotein concentrations. In particular, the increase in postprandial RLP-TG from fasting RLP-TG contributed to approximately 80% of the increase of postprandial total TG from total fasting TG (Table 2). The greater predictive value of non-fasting TG levels associated with cardiovascular events is directly correlated with the increased levels of remnant lipoproteins in the postprandial state.

Serum levels of lipids and lipoproteins and ratios after oral fat load in 54 healthy Japanese male volunteers (mg/dL)
As shown by Stanhope et al.[66] in Fig. 3 in a fructose and glucose treatment study, the significant increase in plasma TG levels during the day was associated with food intake. It was only in the early morning that TG levels in all cases returned to the basal levels. Fig. 3 shows that the TG levels in generally healthy volunteer plasma were the highest at 2 AM in the morning, indicating that postprandial conditions continue even past midnight during the course of a day, except in the early morning. Remnant lipoprotein levels increased significantly during most of the day, except for in the early morning, reflecting a similar profile to that of TG. These increases may depend on the kind of foods. The typical carbohydrate-rich Japanese meal did not increase the levels of TG and remnants during a given day compared with a fat-rich meal such as in the typical Western diet[67], as shown by Ai et al.[68] and Sekihara et al.[69].

The 24-hr circulating plasma TG concentrations in subjects before and after 2, 8, and 10 wk of consuming fructose-sweetened beverages (n = 17). The significant increase in plasma TG levels during the day was associated with food intake in a fructose treatment study. The TG levels in all cases returned to the basal levels only in the morning, and were highest in the middle of the night (Stanhope et al. J Clin Invest 2009;119:1322-34).
Other postprandial studies have been conducted by the oral fat load test. One typical study performed in our laboratory was carried out in Japanese volunteers who were generally healthy with no apparent disease[70]. All participants performed an oral fat tolerance test (OFTT) as previously reported[68,71]. Blood samples were drawn before and 2, 4, and 6 hr after an oral fat load. Plasma apoB-48 level was significantly increased and correlated with the TG levels in postprandial plasma, however, apoB (more than 98% apoB-100) level did not. These results suggest that CM or CM remnants carrying a large amount of TG may be the major component of the increase in the postprandial remnant lipoproteins. However, the apoB100 concentration is known to be much larger than the apoB48 in the plasma. The apoB100 in LDL decreased during a fat load, which resulted in there being no change in apoB despite the increase in apoB100 in the postprandial RLP.
Triglyceride-rich lipoprotein remnants in plasma as a risk factor for cardiovascular disease independent of TG level in plasma
Patients at increased risk of CAD frequently have an atherogenic lipoprotein profile characterized by elevated plasma TRL levels, a predominance of small, dense LDL and reduced HDL cholesterol, which are highly associated with the characteristics of metabolic syndrome. This profile is often seen in patients with type 2 diabetes mellitus with normal LDL concentration and it is associated with an approximately three-fold increase in risk of atherosclerotic disease[72]. An elevated remnant lipoprotein concentration determined as remnant-like lipoprotein particle-cholesterol (RLP-C) is also a characteristic feature of patients with this atherogenic lipoprotein profile and there is considerable evidence linking increased plasma RLP-C levels with CAD[73]. In this connection, Nakajima et al.[8] reported that plasma RLP-C levels were abnormally high in Japanese patients with coronary heart disease. Similarly, Ikewaki et al.[74] showed that plasma RLP-C was significantly increased in postprandial state in patients with coronary artery disease. Further, Leary et al.[75] subsequently found that CAD patients (n=151) from nine centers in the United States and one in Canada had significantly higher median RLP-C levels compared with 302 gender- and age-matched control subjects. Devaraj et al.[76] also showed that RLP-C levels were markedly higher in CAD patients compared with healthy control subjects (P<0.01). These results are supported by other case-control studies reporting that RLP-C levels were significantly high in patients with CAD, patients with resterosis after percutaneous transluminal coronary angioplasty[77,78] in vasospastic angina[79,80], coronary artery stenosis[81], coronary artery endothelial dysfunction[82,83], sudden cardiac death[84,85], intermittent claudication[86], increased intima-media thickness of the carotid artery[87] and in CAD patients with normal cholesterol or TG levels[88,89].
In a large study, NcNamara et al.[90] measured RLP-C and RLPTG in fasting plasma samples from 1,567 women in the Framingham Heart Study. Multiple logistic regression analysis adjusting for other major risk factors (e.g., age, hypertension, smoking, diabetes, LDL-C, HDL-C beta-blocker use and replacement hormones) revealed that RLP-C was an independent risk factor for cardiovascular disease (CVD) in these women and was independent of TG (Table 1). RLP-C has similarly been shown to be an independent risk factor for CAD in Korean patients with type 2 diabetes[91] and in Japanese patients over 65 yr of age[92].
More recently, prospective data have been presented supporting the prognostic value of RLP-C measurement. Three studies have been reported by Kugiyama et al. at Kumamoto University Hospital in Japan, in which CAD patients (men and women[93], postmenopausal women[94] and type 2 diabetes patients[95]) who had angiographically documented arterial stenosis were investigated. Their lipid and RLP-C levels were measured at baseline and then the patients were followed for 2 to 3 yr until the occurrence of a clinical event (recurrent or refractory angina pectoris requiring coronary revascularization, nonfatal myocardial infarction, or cardiac death). In all three of these studies, higher RLP-C levels were associated with greater probability of a coronary event and were found to be independent risk factors (other than the hitherto known risk factors like age, gender, smoking, hypertension, triglycerides, cholesterol, and HDL-C) and were shown to be statistically significant predictor of future coronary events. Recently, high plasma levels of RLP-C have been reported in metabolic syndrome as a risk for endothelial dysfunction and coronary artery disease[96-99].
Analytical approaches: Isolation of RLP using specific antibodies and the diagnostic characteristics of the RLP-C assay
A method for the isolation of TRL remnants according to apolipoprotein content and immune-specificity has been developed to provide quantitative and clinically applicable measurements of plasma remnant lipoproteins as shown in Fig. 4[8,9]. This is the first method to separate remnant lipoproteins from TRL using specific antibodies so as to isolate the remnant fraction under moderate conditions. In this assay, RLP is isolated from plasma in the unbound fraction by immunoaffinity chromatography with a gel containing an anti-apoAI antibody and a specific anti-apoB-100 monoclonal antibody (JI-H) (JIMRO II, Otsuka, Tokyo). The former antibody recognizes all HDL and any newly synthesized CM containing apoA-I, whereas the latter antibody recognizes all apoB-100-containing lipoproteins, except for certain particles enriched in apoE. The anti-apoB-100 antibody JI-H recognizes the B51 region of apoB100 and CM has no epitope in this region. Therefore, CM lacking apoA-I, which is defined as the CM remnants[16], is not recognized by the gel and all such apoB-48 particles in the plasma are isolated in the unbound RLP fraction. The reason the anti-apoB-100 antibody does not recognize the apoE-enriched RLP is not entirely clear, although the amino acid sequence of the epitope region of the apoB-100 antibody is homologous to an amphipathic helical region of apoE, which suggests that apoE should be able to compete for binding of the antibody to its epitope, located between amino acids 2270-2320 from the N-terminal end of apoB-100[9]. The amphipathic helical peptide (2293-2301) of the chemically synthesized antibody epitope reacted with the anti apoB-100 antibody (JI-H) and exhibited potent reverse cholesterol transport activity as apoA-I, apoE or HDL.
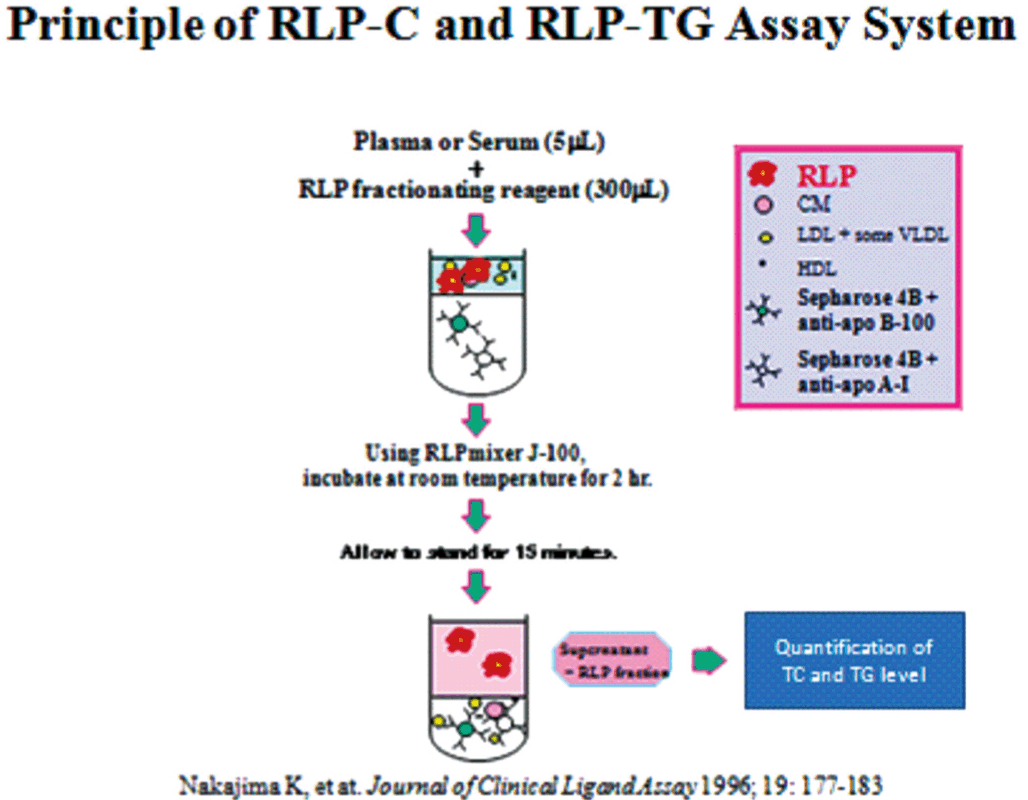
The principal of the RLP-C and RLP-TG assay system. Two specific monoclonal antibodies (anti-apoB100 and anti apoA1) were used to delete plasma nascent VLDL, LDL and HDL and isolate remnant lipoproteins as an unbound fraction by immuno-affinity gels. Cholesterol or TG in the unbound fraction was defined as RLP-C or RLP-TG.
HDL, LDL, large CM and the majority of VLDL are thus retained by the gel. The unbound RLPs comprise remnant VLDL containing apoB-100 and CM containing apoB-48, which are routinely measured in terms of cholesterol, although they can also be quantified in terms of triglycerides or specific apolipoproteins (i.e., apoB, apoCIII, or apoE)[41,42]. The plasma concentration of RLP-C has been shown to be significantly correlated with the plasma concentration of total TG, VLDL-TG, and VLDL-C. It has not been significantly correlated with LDL cholesterol or LDL apoB[75,100].
The physical and chemical properties of lipoproteins that are not recognized by the apoB-100 monoclonal antibody JI-H, subsequently isolated by ultracentrifugation at a density 1.006 g/mL, have been described[40]. These lipoproteins contained more molecules of apoE and cholesteryl esters than those that were bound, consistent with them being remnant lipoproteins. They had slow pre-b electrophoretic mobility compared with the bound VLDL fraction and ranged in size from 25 to 80 nm. Other lipoproteins, however, may be present when the JI-H monoclonal antibody (together with an anti–apoAI antibody) is used to isolate RLP by immunoaffinity chromatography from total plasma in the absence of ultracentrifugation[8,9]. HPLC analysis of RLP fractions isolated in this way from normolipidemic and diabetic subjects[9], and fast protein liquid chromatographic analysis of RLP from type III and type IV patients[42] have revealed considerable size heterogeneity in RLP, with particles ranging in size from VLDL to LDL.
Treatment for cardiovascular disease; fibrates and EPA as representative therapeutic drugs
Fibrates significantly decrease plasma remnant lipoproteins, especially RLP-TG more efficiently than RLP-C
Fibrates can be very useful in managing hypertriglyceridemia through the effect of fibric acid derivatives on postprandial lipemia. Fibrates decrease the production of and enhance the catabolism of triglyceride-rich lipoproteins through the activation of peroxisome proliferator-activated receptor-alpha[101]. Results of clinical studies with fibrates have confirmed their action in decreasing postprandial triglyceride levels by increasing lipoprotein lipase activity, decreasing apolipoprotein CIII production, and by increasing fatty acid oxidation in the liver. It is concluded that fibrates are effective agents in lowering the postprandial increase in remnant lipoprotein particles and retinyl palmitate[102]. Furthermore, fibrates can also affect the postprandial lipid profile by increasing hepatic lipase levels and in some cases, by reducing cholesteryl ester transfer protein activity. The main goal of fibrate therapy is to improve fasting hypertriglyceridemia, which is essential to improving postprandial lipemia[103]. Fibrates are well tolerated by patients and adverse effects have been reported rarely after their administration. In conclusion, bezafibrate therapy decreased RLP-C levels more effectively than pravastatin, and a decrease in RLP-C levels may be associated with a reduction in CV events in CAD patients with high RLP-C levels[104-110].
EPA is one of the factors that may prevent sudden cardiac death associated with elevated remnant lipoproteins in people consuming more fish
Eicosapentaenoic acid (EPA), a major component of fish oil, has the following various effects: anti-platelet aggregation, vasodilation, anti-inflammation, and hypotriglyceridemia, and therefore has potential anti-atherosclerotic effects. The effect of EPA on serum RLPC concentrations in patients with diabetes mellitus was measured in 10 patients with non-insulin-dependent diabetes mellitus[111]. The patients were treated with 900-1,800 mg EPA ethyl-ester daily for 3 mon. Serum RLP-C concentrations and plasma fatty acid composition before and after the administration of EPA were determined. Serum RLP-C concentrations were significantly decreased 3 mon after the administration of EPA. Plasma EPA concentrations and the ratios of EPA to arachidonic acids (AA) were significantly increased during the same period. Serum RLP-C concentrations were inversely correlated with the ratios of EPA to AA in plasma. These results suggest that administration of EPA is effective for remnant hyperlipoproteinemia, which is a risk factor for atherosclerosis and sudden cardiac death[112-116]. In conclusion, EPA administration proved to be an effective and safe treatment to decrease plasma remnant lipoproteins and prevent in vivo peroxidation of LDL in dialysis patients.
Conclusion
The immuno-separation method made it possible to specifically isolate remnant lipoproteins from plasma as an unbound fraction to the anti-apoB100 and apoA-1 immuno-affinity gel. This technique provides a tool for investigating the biochemical and physiological characteristics of remnant lipoproteins in plasma, which show high similarity with ox-LDL as oxidized lipoproteins without additional oxidation in vitro. Also, a new remnant assay was developed to measure the cholesterol in remnant lipoproteins (RLP-C) for clinical diagnostic purposes and demonstrated that RLP-C is an independent risk factor of CHD risk. New insights into remnant lipoproteins as a phoscausative factor for atherosclerosis and thrombosis have been revealed using this antibody separation technique.
Acknowledgements
The author deeply thanks Dr. Richard Havel at University of California San Francisco and Dr. Ernst Schaefer at Tufts University for their long-term collaboration on remnant lipoprotein research.